What Is Sodium Polyacrylate?
Sodium polyacrylate is a synthetic polymer used in cosmetics and skincare products. It is used to improve the sensory feel and stability of products. It functions as a thickening agent, texture enhancer, film-forming agent, and emulsion stabilizer.
Sodium polyacrylate, also known as waterlock, is the sodium salt of polyacrylic acid, a high molecular weight polymer. A polymer is a substance which has a molecular structure built up from a large number of similar units, called monomers, bonded together. With sodium polyacrylate, the monomers are acrylic acid. Functionally, this structure is strong and flexible.
In its dry form, sodium polyacrylate exists as a white, fluffy powder. When it is added to water, however, it turns into a gel-like substance, absorbing many times its weight in water. This is why sodium polyacrylate is considered a “super absorbent polymer”.
The origins of super absorbent polymer chemistry trace back to the early 1960s when the U.S. Department of Agriculture developed the first super absorbent polymer materials. In addition to use in cosmetics, super absorbent polymers have a wide array of applications, including candles, wound dressings, hot and cold therapy packs, diapers, potting soil, and many more.
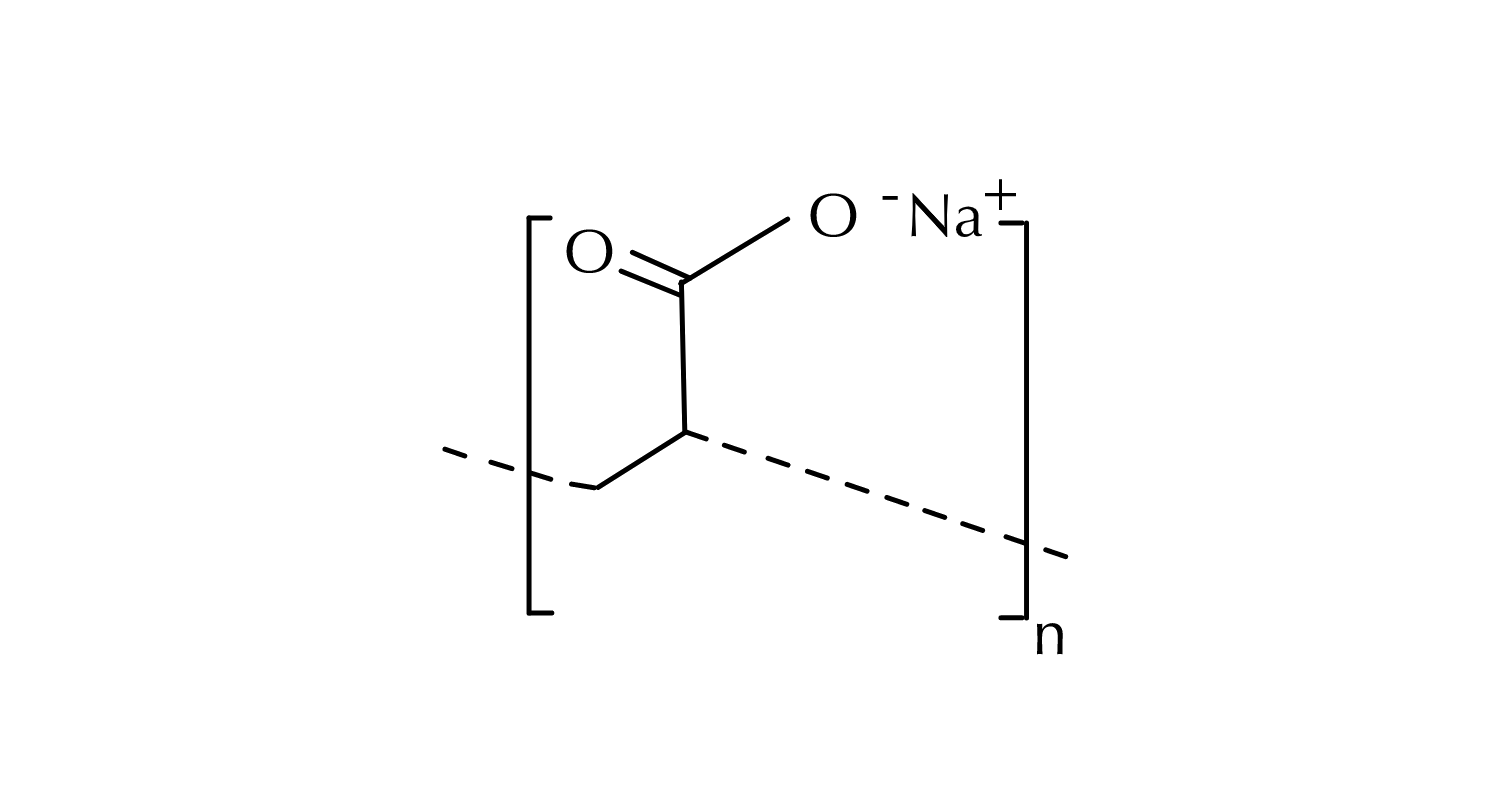
Sodium Polyacrylate
the good:Sodium polyacrylate’s main function is to improve the sensory feel and stability of a product. This means that it helps the product feel smooth and pleasant to use as well as stabilizing the product so that it doesn’t separate or split.
the not so good:Sodium polyacrylate doesn’t cause any irritation or sensitivity.
Who is it for?All skin types except those that have an identified allergy to it.
Synergetic ingredients:Works well with most ingredients
Keep an eye on:Nothing to keep an eye on here.
Why Is Sodium Polyacrylate Used?
In cosmetics and skincare products, sodium polyacrylate functions as a thickening agent, texture enhancer, film-forming agent, and emulsion stabilizer.
Thickness
Sodium polyacrylate functions as a thickening agent based on its ability to absorb as much as 100 to 1000 times its mass in water.
In water-based solutions, the sodium ions in sodium polyacrylate are free to move since they are replaced by positively charged hydrogen ions. This means that instead of an organized polymer chain, a gel forms. As the gel absorbs water it helps to thicken the formulation.
The thickness of a product is an important part of the formulating process as it helps to improve the feel of the cream or lotion. Not only does it affect how the product feels, it also helps to improve how the product works. When a formulation has been thickened it can improve both how evenly the product is spread across the skin and how easy the product is to spread.
Texture
As a texture enhancer, sodium polyacrylate contributes to the smooth, glossy appearance of products. It keeps the texture of a product soft and supple while also conditioning the skin.
By adding sodium polyacrylate to things like shampoos, conditioners, creams, and lotions, formulations will look and feel more rich, smooth, and creamy.
Additionally, sodium polyacrylate can be used in exfoliating products to make strong scrubbing materials more gentle.
Film forming
Sodium polyacrylate is also a film-forming agent. Upon application, sodium polyacrylate creates a thin, flexible film that gives a smooth feeling on the skin and helps to protect the skin after it has been washed.
Stabilizer
Sodium polyacrylate has a stabilizing effect on emulsions. Emulsions are the name we give to formulations that have both water and oil-based ingredients. Often these kinds of products can be hard to keep from separating or splitting, where the oil and water-based ingredients repel eachother.
When water and oil- based ingredients are mixed they create a dispersion of droplets such as oil droplets in water. However, when mixing stopsthe two ingredients can begin to separate.
An emulsifier like sodium polyacrylate can be added to help the droplets remain dispersed.
This improves the consistency of a product, which enables an even distribution of topical skincare benefits.
Is Sodium Polyacrylate Vegan?
Sodium polyacrylate is a vegan ingredient. It is usually synthetically derived.
If you are looking for a vegan product it is best to hceck that the other ingredients in the formulation are also vegan and that the brand is cruelty-free.
Is Sodium Polyacrylate Safe?
The safety of polymers that contain the acrylic acid monomer such as sodium polyacrylate, has been assessed by the Cosmetic Ingredient Review Expert Panel.
The CIR Expert Panel is responsible for the independent evaluation of skincare and cosmetic ingredients. The Expert Panel evaluated the scientific data and concluded that sodium polyacrylate was safe for use in cosmetics and personal care products when formulated to avoid skin irritation.
Since sodium polyacrylate is a large compound, it is unable to penetrate the skin’s barrier. If a compound cannot penetrate through the skin, it cannot react with cells of the immune system. Therefore, sodium polyacrylate does not cause sensitivity reactions.







