What Is Hexylene Glycol?
Hexylene glycol is an ingredient used in skincare and cosmetic products to help improve the texture and sensory feel of the formulation. Hexylene glycol functions as a surfactant, emulsifier, and a viscosity-reducing agent.
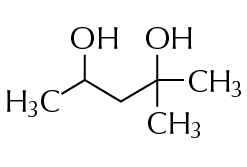
Hexylene glycol, also known as 2-Methyl-2,4-pentanediol, is an organic compound that can be classified as a glycol. Glycols are a class of alcohols that contain two hydroxyl groups, which can also be called a diols. Hexylene glycol is a clear, hygroscopic liquid with a mild, sweet odor.
Hexylene glycol is most often synthetically produced. It is manufactured by the condensation of 2 molecules of acetone to produce diacetone alcohol, which is further hydrogenated to produce hexylene glycol. This is then purified.
Hexylene glycol is used in the formulation of hair and bath products, eye and facial makeup, fragrances, personal cleanliness products, and shaving and skin care products at concentrations ranging from 0.1% – 25%.
Hexylene glycol as a surfactant helps to cleanse and moisturize the skin as well as thinning out the formulation to improve absorption, texture and allow other ingredients to work better.
Hexylene Glycol
the good: Helps to improve the texture and feel of skincare and cosmetic formulations. It also has an added benefit of acting as a protective barrier for the skin.
the not so good: Hexylene glycol can be a mild irritant in high concentrations.
Who is it for? All skin types except those that have an identified allergy to it.
Synergetic ingredients: Works well with most ingredients.
Keep an eye on: Nothing to keep an eye on here.
What Are The Functions Of Hexylene Glycol?
Hexylene glycol is added to cosmetics and skincare products based on its functions as a surfactant, an emulsifier, and a viscosity-reducing agent.
Surfactant
Surfactant is the short term for surface active agents. Surfactants are compounds that lower the surface tension between two substances.
In skincare products, surfactants work to lift dirt, oil and fats from the skin, allowing them to be washed away. This is possible because while one end of the surfactant molecule is attracted to water, the other end is attracted to oil. Thus, surfactants attract the oil, dirt, and other impurities that have accumulated on your skin during the day and wash them away.
Due to these properties, hexylene glycol can be found in many different cleansers and body washes.
Emulsifier
Hexylene glycol also functions as an emulsifier. An emulsifier is needed for products that contain both water and oil components, for example, when oils are added to a water-based formula.
According to EFEMA, when water and oil are mixed together and vigorously shaken, a dispersion of oil droplets in water is formed. When shaking stops, however, the two phases can start to separate. To address this problem, an emulsifier like hexylene glycol can be added to the system, which helps the droplets remain dispersed and produces an even and stable formulation.
As an emulsifier, hexylene glycol consists of a water-loving hydrophilic head and an oil-loving hydrophobic tail. The hydrophilic head is attracted to the water and the hydrophobic tail to the oil. Once again, hexylene glycol reduces the surface tension by positioning itself between the oil and water, which has a stabilizing effect on the product.
Thinning
Finally, hexylene glycol functions as a viscosity-reducing agent. The term viscosity corresponds to the concept of “thickness”, for example, honey has a higher viscosity than water. As a viscosity-reducing agent, hexylene glycol works to thin out heavy formulations and create a thinner, more spreadable product.
Is Hexylene Glycol Vegan?
Hexylene glycol is considered to be a vegan ingredient as it is made synethically made from oils and natural gas.
If you are looking for a vegan product, always make sure that the other ingredients in the product are vegan and that the brand is cruelty-free.
Is Hexylene Glycol Safe?
The safety of hexylene glycol has been assessed by the Cosmetic Ingredient Review Expert Panel. The Cosmetic Ingredient Review Expert Panel is responsible for the independent evaluation of the safety and efficacy of skincare and cosmetic ingredients. The Expert Panel evaluated the scientific data and concluded that hexylene glycol is safe for use in cosmetics and personal care products.
Despite the approval by the CIR Expert Panel, hexylene glycol can be an irritant, especially at higher concentrations, usually outside the scope of skincare.
As a class, the glycols can produce mild to severe ocular irritation with hexylene glycol producing severe irritation at high concentrations.
According to Cosmetics Info, a number of product formulations containing these glycols at concentrations up to 21.4% have been tested in various human skin irritation and sensitization assays. The degree of irritation produced depended upon the particular product.
Furthermore, there was no correlation between the degree of irritation and the concentration of the glycol present in the product. There were no reactions indicative of skin sensitization to these glycols in any skin sensitization and no suggestions of phototoxicity or photosensitization.
A study published in the journal Contact Dermatitis examined the effects of hexylene glycol when used by people with eczema and concluded that the presence of hexylene glycol did not cause a significant increase in irritation.







