What Is Cyclohexasiloxane?
Cyclohexasiloxane is a common ingredient in skincare and body care products, used to improve the texture of formulations. While the texture is usually a sensory aspect of the product, in the case of cyclohexasiloxane, it helps the product to be evenly distributed so the key ingredients can improve the skin. It also has the added benefit of acting as a protective barrier to the skin, protecting the skin from moisture loss, allergens, and bacteria.
Cyclohexasiloxane is a type of silicone. Silicones are synthetic polymers with a backbone composed of repeating units of siloxane, which is why silicones may also be referred to as polysiloxanes. Siloxane is made up of elemental silicon and oxygen. Often, the terms “silicone” and “silicon” are mistakenly used interchangeably, when they are quite different. Silicon is the 14th element on the periodic table and the second most abundant element in the earth’s crust after oxygen. In contrast, silicones are always synthetically produced.
Cyclohexasiloxane is part of a family of small silicones called cyclomethicones. Cyclomethicones are clear, odorless liquids utilized in skincare products to give your skincare a smooth texture that allows the product’s key ingredients to be delivered evenly to the skin. Cyclomethicones are part of the silicone family and are used in a similar way to dimethicone, which you may be familiar with. Cyclomethicones help smooth the texture of products, improve the appearance of scarring, increase the water-resistance abilities of formulations, and emulsify the product. Cyclomethicones are also better at delivering active ingredients to the skin as they are more volatile than larger silicones and slowly evaporate from the skin. When cyclomethicones disappear, they leave behind the formulation’s key ingredients.
Cyclomethicones are also known as cyclosiloxanes, different names for the same class of molecules. The two most commonly used cyclomethicones are cyclohexasiloxane and cyclohexasiloxane; you may have seen these ingredients somewhere near the bottom of your product’s ingredients list.
Cyclohexasiloxane’s structure forms a ring which makes it more volatile or less stable. This instability allows cyclohexasiloxane to evaporate when applied to your skin readily. This characteristic makes cyclohexasiloxane a great ingredient for improving the spreadability of products and ensuring they don’t remain sticky after application. Cyclohexasiloxane gradually evaporates from the skin, leaving behind the other key components in the product to improve the appearance of the skin. This action of evaporation makes it an excellent carrier ingredient.
12 Skincare Ingredients You May Want to Avoid
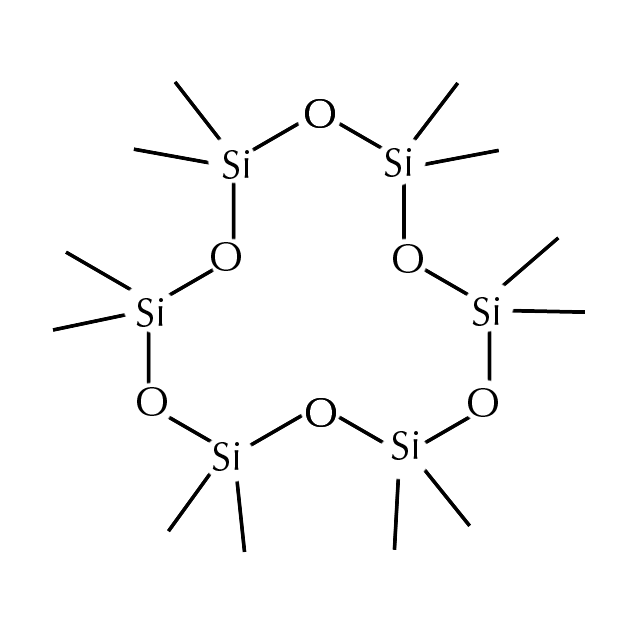
Cyclohexasiloxane
the good: Cyclohexasiloxane helps to improve the texture of products, enhancing their spreadability, and helping to maintain the skin’s natural barrier and protect the skin from moisture loss.
the not so good: Often will need to be combined with heavier silicones to help improve the thickness and spreadability of the product.
Who is it for?All skin types except those that have an identified allergy to it.
Synergetic ingredients: Works well with most ingredients
Keep an eye on: Other names such as Dodecamethylcyclohexasiloxane
What Are the Benefits of Cyclohexasiloxane?
Cyclohexasiloxane, as a silicone, improves the feel, appearance, and performance of skincare and cosmetic formulations. Cyclohexasiloxane is a multifunctional ingredient in skincare and cosmetic formulations.
Texture
Cyclohexasiloxane helps to produce an even, smooth formulation that allows the product to spread evenly over the skin. It prevents the product from catching on dry skin patches and evens skin tone by gliding over pores and wrinkles. It ensures that all areas of the skin are receiving the key ingredients in your product.
Lightweight
Cyclohexasiloxane is a volatile substance due to its ring-shaped structure. This allows it to evaporate from the skin, leaving behind the product’s ‘active’ ingredients to work their magic. It also helps to reduce the heaviness of other silicone ingredients.
Synthetic
Synthetic ingredients often have a bad reputation in the skincare world as they are considered not natural. This plays into the idea that natural is always better, which isn’t always true. Synthetic ingredients are usually safer than their plant or animal-derived counterparts as they contain fewer impurities and are less complex, making them less likely to irritate. They are also better for the environment as it doesn’t deplete a natural resource.
Hydration
Cyclohexasiloxane, along with other cyclomethicones, were analyzed in a study published in Skin Pharmacology and Physiology. The study investigates the occlusive characteristics of the cyclomethicones. Occlusive substances work by creating a physical barrier between the skin and the air, keeping moisture in and allergens and bacteria out.
The study found that cyclomethicones are non-occlusive ingredients due to their volatility. The ability to evaporate from the skin allows cyclomethicone to form a protective barrier for a short time. This is great for congestive skin types as it prevents sweat and skincare ingredients from being pushed deeper into the pores.
However, occlusive barriers are generally useful for drier skin types as they prevent moisture or water loss from the skin’s top layers. Cyclomethicones may still be beneficial for improving the appearance of dry skin as the rate at which the evaporation occurs provides the skin is slow, and the ingredients will still offer the skin with the protective barrier for some of the time it is on the skin.
Who Should Use Cyclohexasiloxane?
Cyclomethicone products are suitable for most skin types, including congested, dry, or sensitive skin types, as well as skin types that may have damaged skin barrier integrity.
Congested skin types
Studies have determined that most silicone products, including cyclohexasiloxane, is non-comedogenic or non-clogging, allowing the skin to breathe. Silicones and cyclomethicones do not enter the pores as they are too big to enter the pores. Pores are where the formation of blemishes occur, so silicones are not responsible for breakouts or worsening of congestion. Silicone products may have the potential to worsen congested skin types as they may trap sweat or other skincare products that potentially cause breakouts, closer to the skin. This theory hasn’t been tested but has some validity given the function of silicones.
To avoid this potential side effect, it is best to make sure you cleanse the skin after exercising and to check the ingredients in your other skincare products to see if they could be contributing to the breakouts.
Sensitivity
Cyclohexasiloxane is an emollient that soothes and softens the skin by keeping moisture locked in. This characteristic helps to prevent water loss and maintain the integrity of the skin’s natural barrier. Protecting or preserving the skin’s natural barrier is an essential part of keeping sensitivity issues at bay. Disruption of the skin’s natural barrier is considered to be linked with skin conditions such as eczema, dermatitis, and psoriasis.
If you have sensitive skin, be mindful of the other ingredients in your product that may irritate, such as fragrance, because the cyclohexasiloxane will deliver these ingredients effectively to the skin. In some cases, minor irritation may occur from the use of silicones, but it’s not usually the source of sensitivity in a product.
Is Cyclohexasiloxane Safe?
The safety of the group of cyclomethicone ingredients, including cyclohexasiloxane, has been assessed by the Cosmetic Ingredient Review Expert Panel. The Panel that there is minimal absorption of the ingredient and the available research did not suggest skin irritation or sensitization. The Panel concluded that these ingredients are safe in the present applications and concentration.
Environment
One issue with cyclohexasiloxane is its potential impact on the environment. Cyclohexasiloxane doesn’t readily degrade. This may have a disrupting effect on the environment. As a precautionary measure, the European Union in 2020, limited concentrations of cyclohexasiloxane in products to 0.1%. 0.1% is generally the concentration it is used at, so this is unlikely to affect the formulation of your favorite skincare products if other countries adopt this same policy.
References:
Brown, A & Barot, L, 1986. ‘Biologic Dressings and Skin Substitutes’, Journal of Clinical Plastic Surgery, vol. 13, is. 1, pp. 69-74. Burgess, I, Lee, P & Brown, C, 2008. ‘Randomised, controlled, parallel-group clinical trials to evaluate the efficacy of isopropyl myristate/cyclomethicone solution against head lice’, Pharmaceutical Journal, vol. 280, pp. 371-375. De Paepe, K, Sieg, A, Le Meur, M & Rogiers, V 2014. ‘Silicones as Nonocclusive Topical Agents, Skin Pharmacology and Physiology, vol. 27, pp. 164-171. Disapio, A & Fridd, P, 1988. ‘Silicones: use of substantive properties on skin and hair’, International Journal of Cosmetic Science, vol. 10, is. 2, pp. 75-89. Johnson, W et al., 2012. ‘Safety Assessment of Cyclomethicone, Cyclotetrasiloxane, Cyclohexasiloxane, Cyclopentasiloxane, and Cycloheptasiloxane’, International Journal of Toxicology, vol. 30, pp. 149-227. Puri, N & Talwar, A, 2009. ‘The Efficacy of Silicone Gel for the Treatment of Hypertrophic Scars and Keloids’, Journal of Cutaneous and Aesthetic Surgery, vol. 2 is. 2, pp. 104-106. Van Reeth, I 2006. ‘Beyond Skin Feel: Innovative Methods for Developing Complex Sensory Profiles with Silicones’, Journal of Cosmetic Dermatology, vol.5, is. 1, pp.61-67.







