What Is Benzyl Alcohol?
Benzyl alcohol is an ingredient that is used in cosmetics and skincare products to improve the scent, texture, and to increase the longevity of a formulation.
Benzyl alcohol is a colorless liquid with a mild pleasant aromatic scent. It is produced naturally by many plants and is found in small quantities in foods such as apples, raspberries, strawberries, grapes, peaches, and tea. Benzyl alcohol is found in many essential oils including jasmine, hyacinth, neroli, rose, and ylang-ylang.
In addition to its natural occurrence, benzyl alcohol can be synthetically prepared by the hydrolysis of benzyl chloride using sodium hydroxide. This was discovered in the 1800s during developments in the coal tar and dye industry.
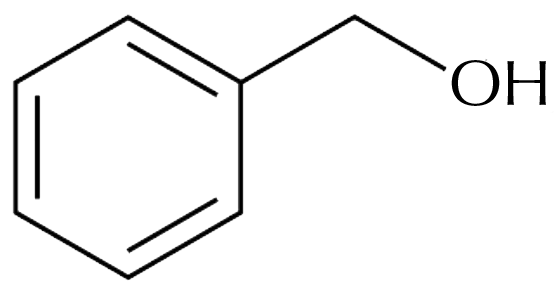
Benzyl Alcohol
the good:Benzyl alcohol is able to act as a preservative and improve the longevity of fragrances as well as improving the texture and sensory feel of products.
the not so good:Nothing to keep an eye on here.
Who is it for?All skin types except those that have an identified allergy to it.
Synergetic ingredients:Works well with most ingredients
Keep an eye on:Look out for the other uses for benzyl alcohol outside skincare.
What Are The Four Main Uses of Benzyl Alcohol?
In cosmetics and skincare products, benzyl alcohol functions as a fragrance component, preservative, solvent, and viscosity-decreasing agent. Most of these functions help to improve the sensory feel and smell of the formulation with little benefit or change to the condition of the skin. While this may make benzyl alcohol seem like an unimportant part of the formulation, it is actually very important to the way the product will last and feel.
Scent
Benzyl alcohol functions as a fragrance ingredient when it is used in high concentrations. Since it is part of the fragrance makeup of essential oils like jasmine, hyacinth, neroli, rose, and ylang-ylang, pure benzyl alcohol imparts a floral-like scent to products. However, as the product ages, the scent can develop changing to a fruity scent and then to an almond scent. The scent of a product is an important component to the product, I’m sure we have all used a product that didn’t quite get the scent right and it made the use rather unpleasant.
Preservative
Benzyl alcohol can also be used as a preservative in cosmetics and skincare products. Benzyl alcohol helps to prevent the growth of bacteria that may be introduced through the air or through contamination through use. The use of preservatives is necessary to prevent bacteria and mold growth, especially since cosmetics have a high potential for contamination. The most susceptible products are creams and lotions that are packaged in jars, opened frequently, and applied to the skin with the fingers. Furthermore, the ingredients used in cosmetic formulations, such as water, oils, peptides, and carbohydrates, plus the warm storage temperatures create the perfect environment for microorganism growth. Microbial contamination can lead to significant health problems, from skin irritation to infections. Preservatives like benzyl alcohol can help to prevent these problems.
As a bacteriostatic preservative, benzyl alcohol works by inhibiting bacterial reproduction, while not necessarily killing them otherwise. This is in contrast to bactericides, which kill bacteria. Since benzyl alcohol cannot kill bacteria, it is typically combined with other preservatives in a cosmetic formulation.
Solvent
Benzyl alcohol also functions as a solvent in cosmetics and skincare products. Solvents help other ingredients dissolve. Solvents can be used to thin out a formulation, which makes the product easier to spread. This is why benzyl alcohol can also be classified as a viscosity-decreasing agent or an ingredient that thins out a formulation. Lastly, solvents can increase the efficacy of active ingredients in a formulation by enhancing their absorption through the skin.
Is Benzyl Alcohol Safe?
The safety of benzyl alcohol has been assessed by the Cosmetic Ingredient Review Expert Panel, a group that evaluates the safety of skincare and cosmetic ingredients. The Panel noted that no adverse side effects of benzyl alcohol were seen in studies that looked at chronic exposure, inhalation, and prolonged skin contact. However, some irritation was noted in some people as such the Expert Panel has approved its use in concentrations up to 5%. Overall, the Panel concluded that benzyl alcohol was safe for use in cosmetic products.
The United States Food and Drug Administration has approved benzyl alcohol as an anesthetic ingredient in over-the-counter anorectal, oral healthcare, and topical analgesic drug products.







