What Is Potassium Cetyl Phosphate?
Potassium cetyl phosphate is an ingredient that is used in a variety of cosmetics and skincare products as a surfactant and emulsifier.
Potassium cetyl phosphate is a potassium salt of a complex mixture of esters of phosphoric acid and cetyl alcohol, a multi-functioning fatty alcohol. It is classified as an alkyl phosphate. The ingredients in the alkyl phosphate family share a common phosphate core structure, and vary by the identity of the alkyl chains, ranging from 8 to 22 carbons in length. Potassium cetyl phosphate has a 16 carbon chain.
Fatty alcohols are a hybrid between alcohols and fatty acids or oils. There is often a misconception that because fatty alcohols have alcohol in their name, that they’re drying to the skin. The reality is actually the opposite.
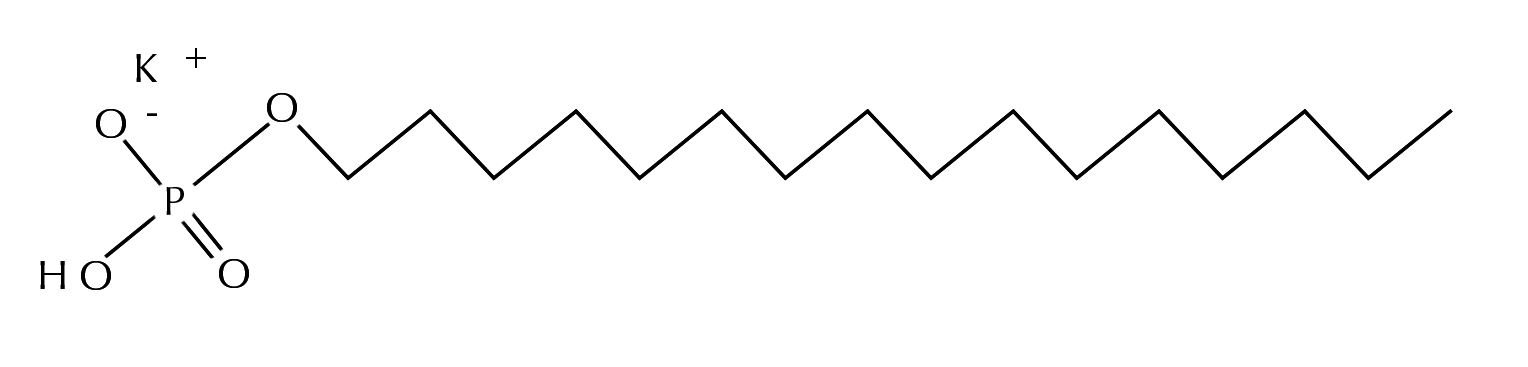
Potassium Cetyl Phosphate
the good:Potassium cetyl phosphate is mainly used to improve the texture and sensory feel of the products that it is used in.
the not so good:It doesn’t provide any direct benefits to the skin and is mainly used to improve the formulation.
Who is it for?All skin types except those that have an identified allergy to it.
Synergetic ingredients:Works well with most ingredients
Keep an eye on:There is some research that may suggest that it can be irritating to some skin types especially those that are already sensitive or have disrupted skin barriers.
Why Is Potassium Cetyl Phosphate Used?
In cosmetics and skincare products, potassium cetyl phosphate functions as a surfactant and emulsifier. It can be found in products such as shampoos, facial cleansers, exfoliants, sunscreens, moisturizers, and makeup removers. According to the FDA’s Voluntary Cosmetic Registration Program data from 2014, potassium cetyl phosphate is reported to be used in 375 formulations, the majority of which are leave-on formulations such as moisturizers, sunscreens and make up removers.
Surfactant
The primary function of potassium cetyl phosphate is as a surfactant. Surfactants are useful cosmetic ingredients because they are compatible with both water and oil. This allows them to lift dirt and oil from the skin and allow it to be easily washed away. This is why potassium cetyl phosphate is used in many cleansing products such as cleansers and shampoos.
Surfactants also function as wetting agents by lowering the surface tension between two substances, such as two liquids or a liquid and a solid. This enables surfactants to spread more easily on the surface, as well as prevent a product from balling up on the surface. This property makes potassium cetyl phosphate a useful ingredient in creams and lotions.
Emulsifier
Another function of potassium cetyl phosphate is as an emulsifier. An emulsifier is needed for products that contain both water and oil-based ingredients. When you mix oil and water-based ingredients they tend to separate and split. To address this problem, an emulsifier like potassium cetyl phosphate can be added to improve the consistency of a product, which enables an even distribution of topical skin care benefits.
Is Potassium Cetyl Phosphate Safe?
The Cosmetic Ingredient Review Expert Panel reviewed the safety of 28 alkyl phosphate ingredients, including potassium cetyl phosphate, as used in cosmetic formulations. The Expert Panel concluded that these ingredients are safe in the present of use and concentration in cosmetics when formulated to be non-irritating.
Despite the approval by the CIR Expert Panel, many medical experts believe that cetyl alcohol, a component of potassium cetyl phosphate, may be irritating for those with sensitive skin. Synthetic fatty alcohols like cetyl alcohol are thought to alter the lipid bilayer of the epidermis and cause allergic dermal reactions. With this information, it is best that those with sensitive skin or skin conditions like eczema, rosacea, or psoriasis perform a patch test with any product containing potassium cetyl phosphate.







