What Is Salicylic Acid?
Salicylic acid is a beta-hydroxy acid that is commonly used in skincare products to help visibly improve the texture of the skin, reduce the signs of aging and increase skin cell turnover, reducing blemishes. Salicylic acid is mainly used in anti-aging and acne products.
Salicylic acid is the most commonly used beta hydroxy acid or BHA in cosmetics and skincare products. BHAs are a class of acids that exfoliate the skin, removing dead skin cells and lifting dirt and oils. This makes BHAs a great ingredient for blemishes or acne as it removes dead skin cells that build up and contribute to breakouts. This same exfoliating effect helps to smooth the appearance of texture and fine lines. As Healthline discussed in their article, salicylic acid is mainly used in acne treatments and products.
Salicylic acid can be obtained from the bark of willow trees. The active extract of the bark is salicin, which is named after the Latin name for the white willow, Salix alba. When used in skincare and cosmetics, salicylic acid is mainly produced
In terms of the chemistry, salicylic acid is an organic compound made up of a carboxylic acid functional group (-COOH) and a hydroxyl functional group (-OH) separated by two carbon atoms. With salicylic acid, both the hydroxyl and carboxylic acid groups are attached directly to an aromatic benzene ring, rather than along a linear carbon chain.
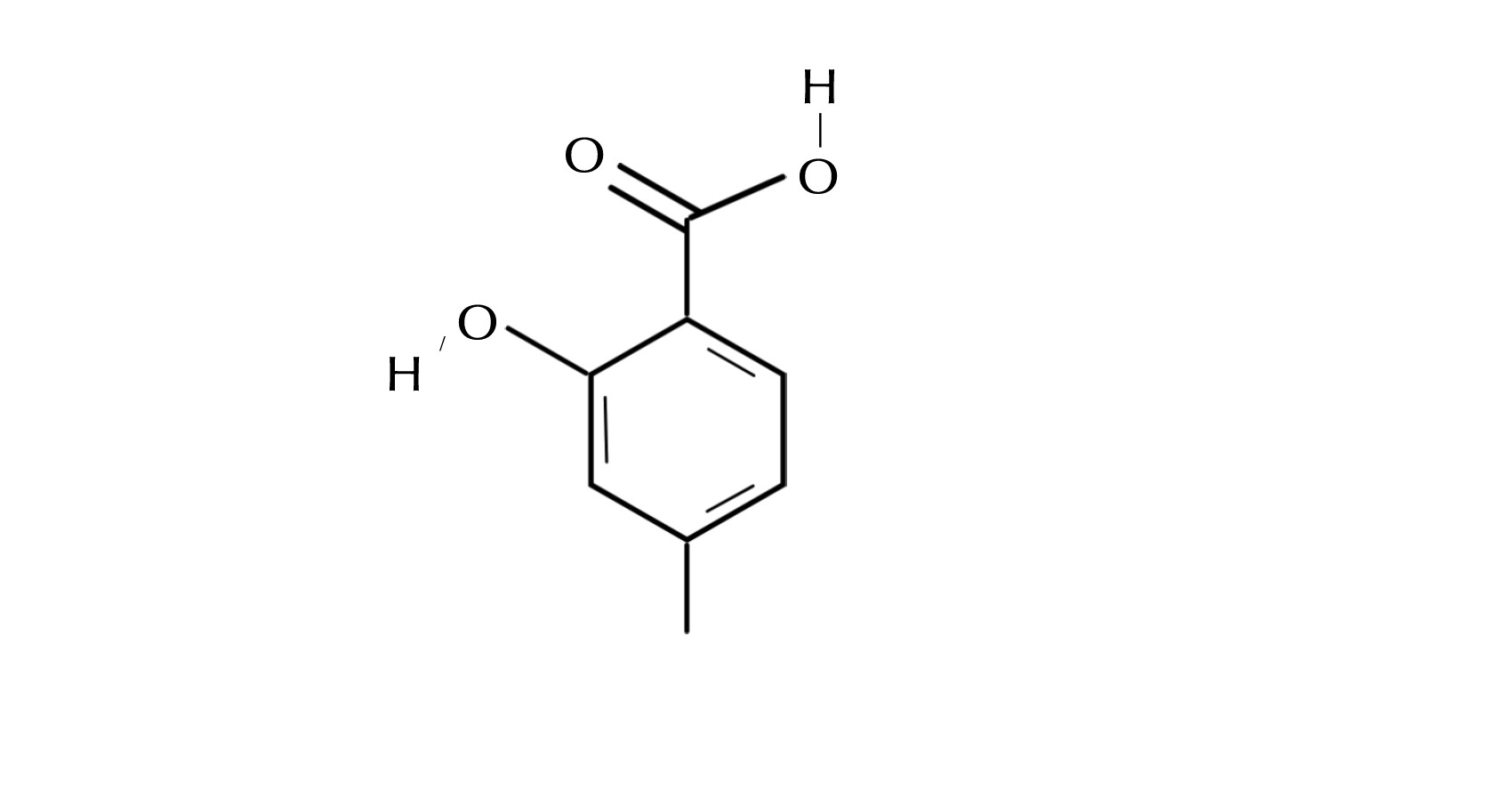
Salicylic Acid
the good:Salicylic acid helps to exfoliate the skin, removing skin cells. This helps to improve the appearance of blemishes and fine lines.
the not so good:Like many acids that are used in skincare and cosmetic formulations, salicylic acid can cause irritation, dryness and flakiness.
Who is it for?All skin types except those that have an identified allergy to it.
Synergetic ingredients:Works well with most ingredients
Keep an eye on:Keep an eye out for irritation and sensitivity.
Why Is Salicylic Acid Used?
Salicylic acid is well known for its ability to help reduce blemishes and acne, which is why it is found in many over-the-counter or OTC acne products. In order to understand how salicylic acid treats acne, let’s first discuss what causes acne.
Acne
The first step in the formation of a blemish involves sebum, a naturally occurring oily substance that moisturizes, lubricates, and protects the skin and hair. While sebum production is a normal process, too much sebum can lead to clogged pores. The next step in the development of acne is triggered when follicles in the skin are not exfoliating properly. This means that keratin, a protein found within the skin, and sebum clog the follicle. When the follicle becomes clogged, a species of bacteria called Propionibacterium acnes accumulate and multiply in the follicle. The follicle becomes inflamed, thus leading to pimple formation.
Considering how acne develops, an effective treatment should work to balance the skin’s sebum levels, reduce the Cutibacterium acnes, formerly Propionibacterium acnes bacteria that contribute to the infection, and reduce inflammation. Studies suggest that salicylic acid works by all three of these actions.
Salicylic acid works well to balance sebum levels because it is lipid or oil soluble. So rather than remaining on the surface of skin, salicylic acid penetrates deep into sebum-filled pores. It then exfoliates the pore lining, which helps to loosen clogged dirt and oil and helps to wash these impurities away. Another way salicylic acid helps to fight acne is through its antibacterial activity. Salicylic acid is thought to have the ability to inhibit the production of various aspects necessary for bacterial reproduction, such as fibrinogen, fibronectin, and alpha-hemolysis. Therefore, salicylic acid may neutralize the bacteria C. acnes that is present in the blemishes. Lastly, salicylic acid is thought to possess anti-inflammatory properties, which is ideal for treating acne since inflammation will worsen breakouts.
Anti-aging
In addition to functioning as an acne treatment, salicylic acid is used in anti-aging skin care products due to its ability to brighten the complexion. It functions as an exfoliant by softening and dissolving keratin, thus allowing dead skin cells to shed naturally. Studies have suggested that the exfoliating properties of salicylic acid may reduce the appearance of fine lines and wrinkles. Over time, exfoliation helps to reduce the thickness of the layer of dead skin cells, giving the skin a brighter, more glowing appearance.
Salicylic acid is approved by the US Food and Drug Administration or FDA for use in OTC drug products. In addition to being FDA approved as a safe and effective acne drug product, it is also approved for use in OTC drugs for corn, callus and wart removal, as well as in anti-dandruff drug products.
Well-documented side effects of products containing salicylic acid include stinging, burning, dryness, peeling, and flaking of the skin. If you have sensitive skin, you should use products with salicylic acid with caution. According to the Mayo Clinic, people using products containing salicylic acid should not apply the following on the same area: abrasive soaps or cleansers, alcohol-containing preparations, benzoyl peroxide, resorcinol, sulfur, tretinoin, cosmetics or soaps that dry the skin, medicated cosmetics, other topical medicine for the skin.
Due to its exfoliative properties, salicylic acid can make the skin more sensitive to sunlight. For this reason, the FDA recommends using a sunscreen containing SPF 30 or more in conjunction with any product containing salicylic acid.
References:
Rendon, M, Berson, D, Cohen, J, Roberts, W, Starker, I, & Wang, B, 2010. Evidence and considerations in the application of chemical peels in skin disorders and aesthetic resurfacing. The Journal of Clinical and Aesthetic Dermatology, 3(7), 32–43.
Soleymani, T, Lanoue, J, & Rahman, Z, 2018. ‘A Practical Approach to Chemical Peels: A Review of Fundamentals and Step-by-step Algorithmic Protocol for Treatment’, The Journal of Clinical and Aesthetic Dermatology, 11(8), 21–28



